New Discovery Reveals Oxygen Flow from Inside Catalysts
Scientists have directly observed the flow of oxygen atoms not only on the surface but also from within catalysts. Researchers at the Dalian Institute of Chemical Physics identified the ‘bulk oxygen spillover’ process using Environmental Transmission Electron Microscopy. This discovery transforms the understanding of catalysts into a three-dimensional concept, potentially enhancing energy production and environmental purification technologies. Kathmandu, April 21.
Challenging longstanding assumptions about catalyst mechanisms, the team from the Dalian Institute of Chemical Physics demonstrated for the first time that oxygen atoms flow from inside the catalyst, not just from its surface. Published in the journal Nature, this research opens new pathways for developing smarter and more efficient catalysts in the future.
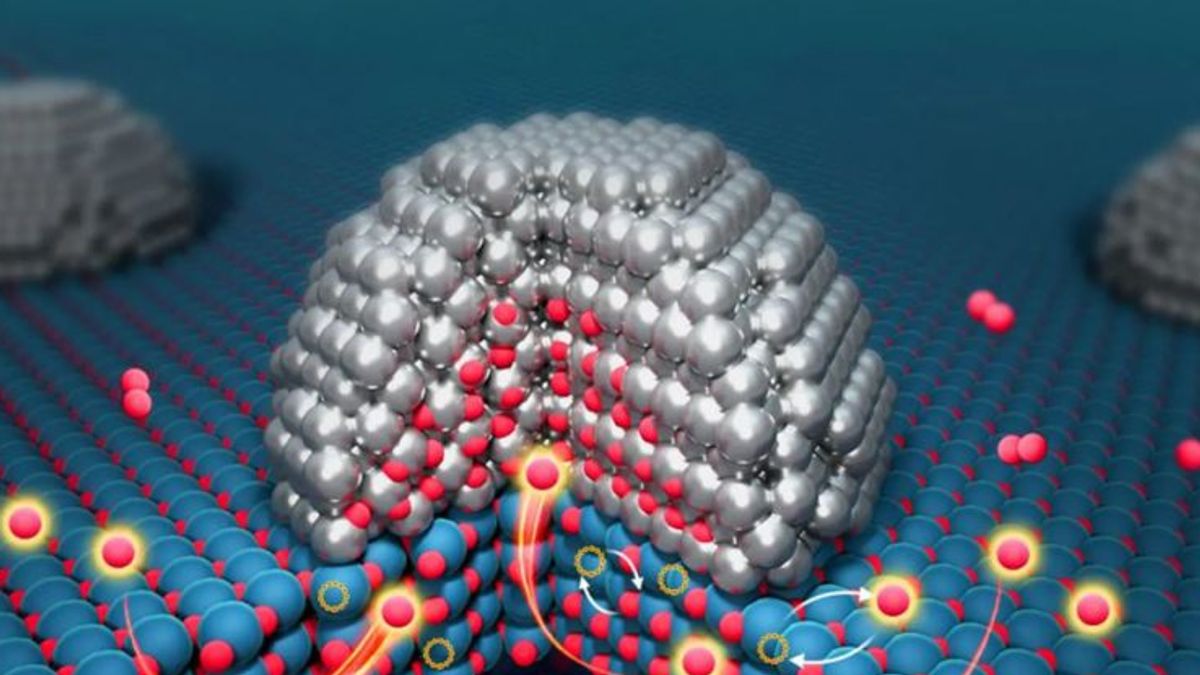
Traditionally, it was believed that only the external surface of catalysts was active in accelerating chemical reactions, with the inner bulk considered inactive. However, under the leadership of Professors Tao Zhang and Yankiang Huang, the researchers used Environmental Transmission Electron Microscopy to track oxygen atoms flowing from the interior layers of the catalyst to the metal surface. They have termed this phenomenon ‘bulk oxygen spillover.’
To understand the role of titanium dioxide, the team experimented with a mixture of ruthenium (Ru) and titanium dioxide (TiO2). Titanium dioxide exhibited significant oxygen storage and release capabilities. During the tests, oxygen atoms were found to flow from 3 to 5 atomic layers beneath the surface to the metal–support interface. According to Professor Wei Liu, the catalyst interface functions as an ‘atomic guard,’ regulating oxygen flow.
This breakthrough challenges the nearly 50-year-old belief that interactions between metals and their supporting materials are limited to the surface. Instead, the new findings redefine catalyst structures from a two-dimensional surface concept to a coordinated three-dimensional system comprising the surface, interface, and bulk interior. This advancement is expected to improve catalyst efficiency notably in energy production, plastic manufacturing, and environmental purification. Professor Zhang emphasized that the next goal is to engage the inner part of catalysts directly in chemical reactions to reduce production costs and enhance functionality.